What Makes Our Platform Different
Phenotypic by Design
There is an urgent need for more rapid tests to identify the causative organisms responsible for bacterial infections as well as drug effectiveness and dose.
Cobio set out to address this by using species-specific phage amplification to measure bacterial viability and antibiotic response, delivering true phenotypic insight rather than inferred resistance based on genetic markers alone.
Naturally Exponential Signal Amplification
Cobio’s approach harnesses the power of natural phage infection and rapid exponential progeny expansion to significantly increase diagnostic signal strength in hours not days.
Readout-Agnostic
The platform integrates with multiple downstream detection modalities, including MALDI‑TOF mass spectrometry, nanopore sequencing, or immunoassay‑based readouts.
Scalable & Adaptable
Because lytic phages exist for most clinically relevant bacterial pathogens, Cobio’s platform can be extended well beyond fixed test panels.
Designed for Real-World Impact
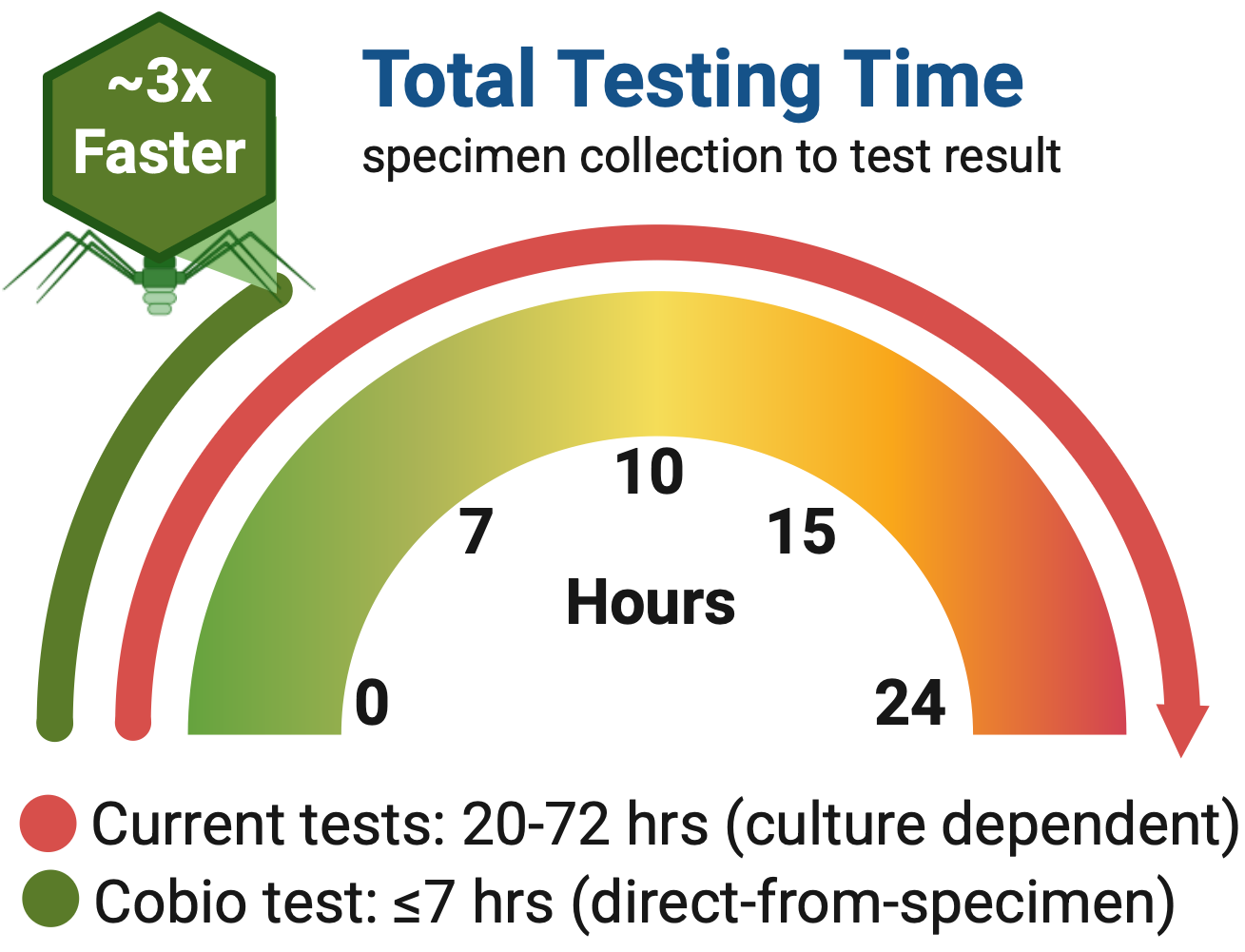
Unlike conventional diagnostics that rely on prolonged single-species cultures (requiring 18-72 hrs) or static molecular signatures, Cobio uses lytic bacteriophages as biological amplifiers. Phage replication occurs only in viable, susceptible bacteria, generating rapid, quantitative signals in as little as 2 hours that reflect real‑time antimicrobial response, even in mixed bacterial specimens. This is especially important because the risk of mortality associated with such resistant infections increases 7% per hour every hour that treatment is delayed.
Cobio’s biological foundation enables same‑shift diagnostic insight while supporting responsible antibiotic stewardship.
Support
Cobio’s research and development efforts are guided by experimental data and informed by ongoing input from experienced diagnostics industry professionals and practicing clinicians.
Cobio’s work is supported by the National Institutes of Health (NIH) and the National Science Foundation (NSF), America’s Seed Fund SBIR Programs, The State of Colorado’s Office of Economic Development and International Trade and the Traxion Venture Network in Golden, CO. All technologies are currently in pre‑commercial development.